Optical Purity, Ibuprofen
Sample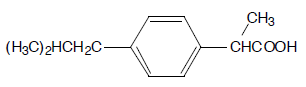
Ibuprofen, racemate (USP reference) and (+) enantiomer
Application
Active ingredient
Conditions
Measuring cell: DSC820
Pan: Aluminum 40 µl, hermetically sealed
Sample preparation: As received, the mixtures are dissolved in a little methanol and left to crystallize slowly for several days at 6 °C
DSC measurement: Heating from 30 °C to 110 °C at 5 K/min
Atmosphere: Nitrogen, 50 cm3/min
Interpretation
Isomers are substances with the same chemical composition but different structure. Enantiomers are isomers which show optical activity. If the (+) and (-) enantiomers are present in the same ratio in a crystal, then it is known as a racemate. Racemates and pure enantiomers have different crystal structures, different solubilities and melting points and can therefore be distinguished from each other by DSC. The (+) enantiomer is, in this case, the pharmacologically active form. Compared with the racemate, it has a greater solubility and a lower melting point. This can have consequences in the formulation of pharmaceutical preparations, if for example, the racemate and enantiomer should be able to be replaced by one another.
Evaluation
The onset temperatures and heats of fusion of the peaks are determined. The onset temperatures are required for constructing the phase diagram.
| Sample | Onset, °C | ΔH, J/g | Onset, °C | ΔH, J/g |
|---|---|---|---|---|
| Racemate | - | - | 74.4 | 121.8 |
| (+) Enantiomer | 50.4 | 86.6 | - | - |
| Mixtures | ||||
| 95% enantiomer / 5% racemate | 47.5 | 81.7 | - | - |
| 90% enantiomer / 10% racemate | 47.6 | 77.4 | - | - |
| 75% enantiomer / 25% racemate | 43.6 | 35.7 | 63.7 | 39.4 |
Note: 25% racemate corresponds to 12.5% (-) enantiomer
The diagram shows the onset temperature as a function of the enantiomer concentration (weight %).
The phase diagram (liquidus curve) shows the onset temperatures as a function of the (-) enantiomer concentration. A concentration of 0% means 100% (+) enantiomer, 50% corresponds to the racemate, and 100% is pure (-) enantiomer. The right section of the diagram (concentration 50% bis 100%) was obtained by forming the mirror image of the left part.
Conclusion
DSC can distinguish between racemates and the pure enantiomers. A contamination of the (+) enantiomer with racemate can be detected. This is observed as a broadening of the melting peak and a depression of the melting point.
DSC can not, however, distinguish between the pure (+) and (-) enantiomers because they have the same melting points. One can, however, add the (+) form to the unknown enantiomer: an unchanged melting peak shows that the unknown substance is also present in the (+) form.
Optical Purity, Ibuprofen | Thermal Analysis Application No. HB825 | Application published in METTLER TOLEDO TA Application Handbook Pharmaceuticals